Introduction to NIFTY® mono
NIFTY® mono is a non-invasive prenatal test for single-gene conditions. It screens for various clinically significant and life-altering genetic conditions in a fetus that are undetectable by current NIPT technology, allowing for a more complete picture of the associated risks when a pregnancy is being adversely affected by a genetic condition.
Some conditions in NIFTY® mono are not typically associated with abnormal prenatal ultrasound findings (especially in the first trimester), or may not be evident until the late second/third trimester or after delivery. Therefore, this test is the next leap in the evolution of screening for genetic disorders during pregnancy, providing valuable information for medical decisions, preparation, and peace of mind for families and physicians.

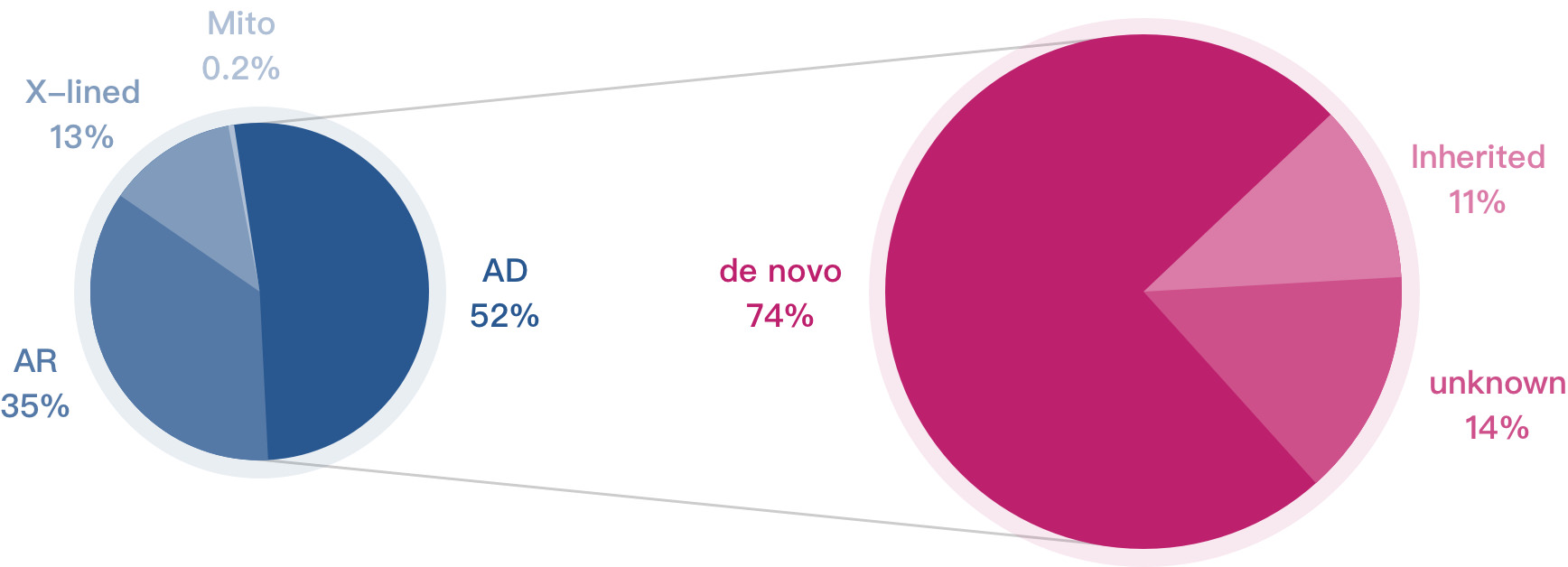
Although the occurrence of dominant single-gene conditions is relatively rare, its cumulative rate is more than 1/1500[1]
Single-gene conditions areresponsible for a heavy loss of life, among which dominant inheritance accounts for more than 50% of all single-gene conditions[2]
74% of dominant single-gene conditions are caused by de novo mutations (a gene mutation that is not inherited)[2]. Family history is typically not a good indicator of probability.
Indication for Testing
NIFTY® mono offers a comprehensive screening for several skeletal, cardiac, and syndrome conditions. NIFTY® mono can provide important information for people who:
• Are of advanced paternal age[3],[4]
• Have ultrasound anomalies, such as shortened long bones and increased NT, which is suggestive of monogenic disorders
• Would like to avoid an invasive diagnostic procedure
• Want to know “everything”
• Are at risk for the genetic conditions being screened for


NIFTY® workflow
10-12 working days after receipt of samples
-
1

Conduct pre-test genetic counseling and ensure patient provides informed consent for test
-
2

Discuss and fill in the NIFTY® Consent Form/Test Request Form with the patient
-
3

Conduct blood draw
-
4

Send scanned copies of Consent Form/Test Request Form and information sheet to BGI
-
5

Arrange collection of the blood sample with a courier
-
6

Send Consent Form/Test Request Form with blood sample to BGI laboratory
-
7

Receive results back in 10 working days
-
8

Conduct pre- and post-test genetic counseling and provide drug guidance advice as required
References:
1.https://www.ncbi.nlm.nih.gov/books/NBK1116/
2.Yang, Y. et al. Molecular Findings Among Patients Referred for Clinical Whole-Exome Sequencing[J]. JAMA, 2014, 312(18): 1870-1879.
3.Toriello H V, Meck J M. Statement on guidance for genetic counseling in advanced paternal age[J]. Genetics in Medicine, 2008, 10(6):457-460.
4.Kong A, Frigge M L, Masson G, et al. Rate of de novo mutations, father’s age, and disease risk[J]. Nature, 2012, 488(7412):471-475.

